T-Cell Activation and Cell Killing Analytics using Laser Force Cytology™
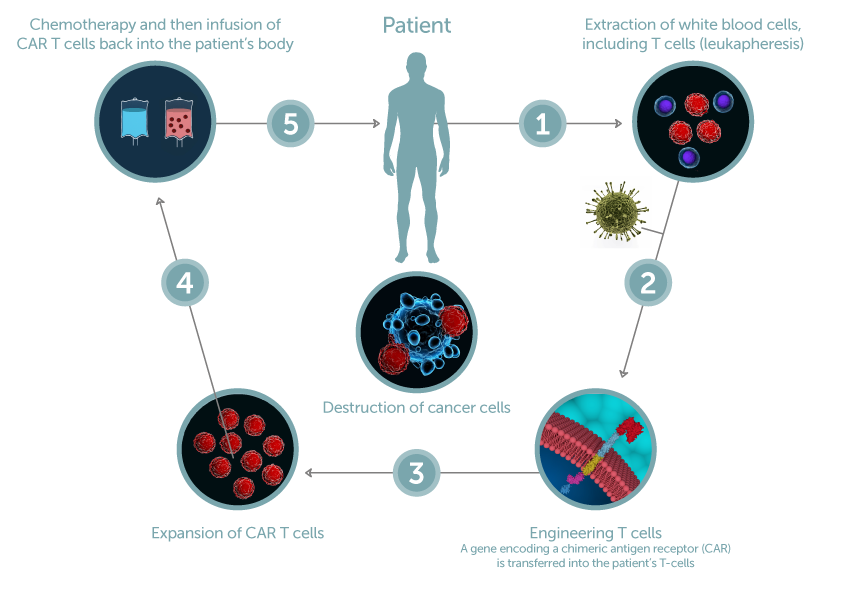
Gene modification cell therapy involves removing cells from a patient and genetically modifying them to introduce a new gene or correct a faulty existing gene. Chimeric antigen receptor (CAR)-T cell therapy is a key example, outlined in the figure on the left. First, T cells are removed from the patient (1) and can undergo pre-manufacturing donor qualification and predictive donor cell analytics to forecast downstream manufacturing success and functional outcomes (1). The cells are then genetically modified through delivery of a new gene using a viral vector (2). This gene modification reprograms the T cells to recognize and attack a certain target cancer cell, thereby promoting specific cancer cell destruction and clearance. The next step in the process is to expand (3) or activate and expand/grow the modified T-cells into a large enough quantity, followed by label-free CAR T cell potency analytics and expansion quality testing (4) prior to infusing the modified cells into the patient (5) to treat their disease, in this case cancer.
Monitoring T-cell activation poses a critical challenge in cell therapies like CAR T therapy, where precise control is paramount. Achieving the right balance in activation is challenging, as overstimulation may lead to exhaustion or adverse effects, while under-stimulation can compromise therapeutic efficacy. Rapid, label-free T-cell activation monitoring is essential for ensuring optimal activation and overcoming a key hurdle in advancing successful cell therapies—Radiance® delivers these insights in minutes, enabling faster decisions and streamlined development.
Using Laser Force Cytology™ to Monitor CQAs and CPPs
Radiance® Laser Force Cytology™ (LFC™) effectively measures intrinsic biophysical and biochemical signatures to track critical quality attributes (CQA) and critical process parameters (CPP), enabling non-invasive cell characterization and near real-time predictive analytics for manufacturing decisions. These label-free cellular fingerprints correlate with expansion quality, potency, and downstream functional success.
Apheresis – Donor Screening and Predictive Analytics
Post apheresis, LFC™ enables predictive donor cell analytics to assess donor variability, replace simplistic viability measures with richer vitality and fingerprint metrics, and predict expansion quality and time from thaw.
Cell Expansion – Real-Time Expansion Quality Monitoring
LFC™ provides label-free CAR T cell analysis during expansion to monitor growth, phenotype shifts, and functional readiness, giving early indicators of expansion quality and likely manufacturing yield.
Quality Control – Cellular Fingerprinting and Potency
LFC™ supports QC with detailed cellular fingerprinting and potency readouts, emphasizing that viability does not equal vitality and that functional, label-free potency metrics better predict therapeutic performance.
Rapid Label-Free Monitoring of T-Cell Activation with Laser Force Cytology™
Here we present LFC™ as a rapid, sensitive method for monitoring T-cell activation that uses optical pressure and microfluidics to measure intrinsic biophysical and biochemical changes in single cells. The advanced label-free CAR T cell analysis metrics measured by LFC™ provide a distinct advantage over label-dependent methods such as flow cytometry and fluorescence-based assays that require fluorescent labeling or other bias-inducing, time-consuming processing steps. LFC™ technology provides rapid, sensitive T-cell activation monitoring and non-invasive cell characterization that reduces bias and sample processing time.


Detection of Mediated CAR T Cell Killing and Potency Assessment
Changes in LFC™ metrics across cell populations quantify donor-specific killing and correlate with CAR T killing potency.
The results demonstrate the capability of Radiance® to measure target-cell killing in a rapid, label-free manner. An LFC™ in vitro assay evaluating CD19 CAR Ts against NALM6 targets shows that measured target-cell loss reflects CAR T potency. Testing multiple Effector:Target ratios reveals donor-dependent killing robustness, with some donors showing consistently higher potency and others lower—demonstrating how predictive donor analytics from Radiance® can guide manufacturing decisions and clinical outcomes.
Accelerate Development and Manufacturing of Life Saving Therapies with Label-Free CAR T Cell Potency Analytics
Harness Laser Force Cytology™ for label-free CAR T cell analysis, predictive donor cell analytics, and rapid T-cell activation monitoring to improve expansion quality, manufacturing success, and final product potency.



